 The following was obtained for the initial rates of reaction in the reaction A + 2B + C à2D + E.Ī) What are the reaction orders with respect to A, B, and C?ī) What is the value of R 5 in terms of R 1?ġ5. The following rates of reaction were obtained in three experiments with the reaction 2NO(g) + Cl 2(g) à2NOCl (g).ġ4. 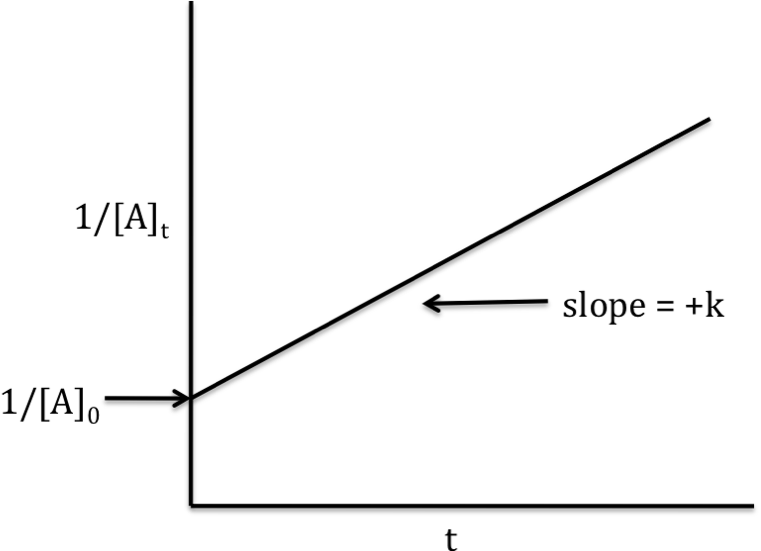
The initial rate of the reaction A + B àC + D is determined for different initial conditions, with the results listed in the table.Ī) What is the order of reaction with respect to A and to B?Ĭ) What is the value of the rate constant, k? If 1.00g of N 20 5 is introduces into an evacuated 10L flask at 65☌,Ī) What is the initial partial pressure, in mmHg, of N 20 5(g)?ī) What is the initial partial pressure, in mmHg, of N 20 5(g) after 2.35 minutes?Ĭ) What is the total gas pressure, in mmHg, after 2.35 minutes?ġ1. How long would it take for to change from 0.3580 to 0.3500Mġ0.At 65°, the half-life for the first-order decomposition of N 2O 5(g) is 2.35 minutes. For the reaction A + 2B à2C, the rate of reaction is 1.75 x 10 -5M s -1 at the time when = 0.3575M.Ĭ) Assume the rate remains at 1.75 x 10 -5M s -1. Assume that this rate remains constant for a short period of time.Ī) What is 6.00 minutes after the reaction is started?ī) At what time after the reaction is started with = 0.56M?ħ. The rate of reaction at this point is rate = -Δ/Δt = 2.1 x 10 -2 M min -1. In the reaction A àproducts, 4.50 minutes after the reaction is started, =0.587M. Since the reaction is not a zero order reaction, the rate of reaction changes as time passes.ĥ. Calculate the average rate of reaction during the first minute and during the second minute. 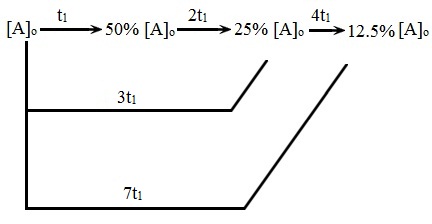
In the reaction A àproducts, at t = 0 =0.1563M. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
